Cdcl2 Crystal Structure

Cdcl2 Crystal Structure. Structure, properties, spectra, suppliers and links for: Atoms form a repetitive pattern called lattice. • have the simplest crystal structures. In this video i provide an overview of the atomic positions within the perovskite or barium titanate crystal structure. Crystal structure determination and refinement gave the x values listed beneath fig. Formed by filling half of octahedral holes in close unit cells are common. Cadmium iodide, cdi2, has a very similar crystal structure to cdcl2. The individual layers in the two structures are identical, but in cdcl2 the. Cdcl2 anion coordination number, shape. The cdcl2 structure may be represented by a hexagonal unit cell, although a smaller rhombohedral cell can also be chosen.
Crystal structure determination and refinement gave the x values listed beneath fig. An „imaginary parallel sided region of a structure from which the entire crystal can be constructed by purely translational cdcl2 cubic close packing. The rcsb pdb also provides a variety of tools and resources. Empirical formula (hill notation) cdcl2. Formed by filling half of octahedral holes in close unit cells are common. Finally, stick the cd in between alternate layers of the chlorine/iodine. The net result for cdcl2, is that the unit cell is indeed rhombohedral, since there is one cd layer for every two cl layers, so it is stretched in that direction. Users can perform simple and advanced searches based on annotations relating to sequence, structure and function. Basic structural chemistry crystalline state structure types. We will look at three such structures.

Structure, properties, spectra, suppliers and links for:
Also known are cdcl2•h2o and cdcl2•5h2o. The individual layers in the two structures are identical, but in cdcl2 the. An „imaginary parallel sided region of a structure from which the entire crystal can be constructed by purely translational cdcl2 cubic close packing. Caf2, tho2, zro2, ceo2 cadmium chloride: • we still describe eutactic structures as ccp or hcp lattices with ions in some fraction of the interstitial sites. When agcl and cacl2 are mixed, each added ion of ca2+ replaces ag+ as they have similar radii. Formed by filling half of octahedral holes in close unit cells are common. Molding porous materials in three dimensions by colloidal crystal templating. Users can perform simple and advanced searches based on annotations relating to sequence, structure and function. Start studying crystal structures (tbc). Cation displacements in perovskites can lead to. Atoms form a repetitive pattern called lattice. • have the simplest crystal structures. The net result for cdcl2, is that the unit cell is indeed rhombohedral, since there is one cd layer for every two cl layers, so it is stretched in that direction. In this video i provide an overview of the atomic positions within the perovskite or barium titanate crystal structure.
Molding porous materials in three dimensions by colloidal crystal templating. Crystal structure determination and refinement gave the x values listed beneath fig. Cadmium iodide, cdi2, has a very similar crystal structure to cdcl2. If a crystal breaks along a plane related to the lattice structure, the plane is called a cleavage plane. Arrays of points (atoms) arranged such that each point has an identical surroundings. •all metals are crystalline solids (special atomic arrangements that extend through out the entire material). Structure, properties, spectra, suppliers and links for: Colourless crystals or white powder ou chemical safety data (no longer updated) more details. We will look at three such structures.
That one mole of cacl2 provides one mole of by ag vaccancies in crystal structure.
These molecules are visualized, downloaded, and analyzed by users who range from students to specialized scientists. The rcsb pdb also provides a variety of tools and resources. Crystal structure determination and refinement gave the x values listed beneath fig. •structures of many inorganic compounds may be initially described in terms of simple packing of half octahedral; Methods of structuring materials with submicrometer features in two dimensions have become highly. Basic structural chemistry crystalline state structure types. Structure plays an important role in understanding properties need to be introduced to different ways of rutile (tio2), cdcl2 and cdi2 structures. • have the simplest crystal structures. Cadmium iodide, cdi2, has a very similar crystal structure to cdcl2. Alternate layers have fully occupied sites. Empirical formula (hill notation) cdcl2. Formed by filling half of octahedral holes in close unit cells are common. Finally, stick the cd in between alternate layers of the chlorine/iodine.
The cube side for silicon is 0.543 nm. Atoms form a repetitive pattern called lattice. Types and crystal structures of normal & inverse spinels are discussed by aditya vardhan v. • we still describe eutactic structures as ccp or hcp lattices with ions in some fraction of the interstitial sites. We will look at three such structures. Start studying crystal structures (tbc). Crystal structure determination and refinement gave the x values listed beneath fig. The rcsb pdb also provides a variety of tools and resources.
Atoms form a repetitive pattern called lattice.
If a crystal breaks along a plane related to the lattice structure, the plane is called a cleavage plane. • unit cell is a simple hexagonal lattice with a. Finally, stick the cd in between alternate layers of the chlorine/iodine. The rcsb pdb also provides a variety of tools and resources. • have the simplest crystal structures. Start studying crystal structures (tbc). Colourless crystals or white powder ou chemical safety data (no longer updated) more details. Learn vocabulary, terms and more with flashcards, games and other study tools. Molding porous materials in three dimensions by colloidal crystal templating. In this video i provide an overview of the atomic positions within the perovskite or barium titanate crystal structure. Fázové přeměny za zvýšeného tlaku. Arrays of points (atoms) arranged such that each point has an identical surroundings.
•all metals are crystalline solids (special atomic arrangements that extend through out the entire material) cdcl2. Also known are cdcl2•h2o and cdcl2•5h2o.
 Source: slideplayer.com
Source: slideplayer.com If a crystal breaks along a plane related to the lattice structure, the plane is called a cleavage plane.
An „imaginary parallel sided region of a structure from which the entire crystal can be constructed by purely translational cdcl2 cubic close packing.
Alternate layers have fully occupied sites.
 Source: chem.libretexts.org
Source: chem.libretexts.org Types and crystal structures of normal & inverse spinels are discussed by aditya vardhan v.
 Source: www.york.ac.uk
Source: www.york.ac.uk • we still describe eutactic structures as ccp or hcp lattices with ions in some fraction of the interstitial sites.
 Source: cdn.rcsb.org
Source: cdn.rcsb.org If a material name (or a mineral name) is clicked, the crystal structure figure which illustrated the atomic (or polyhedron) arrangement of each material.
 Source: c8.alamy.com
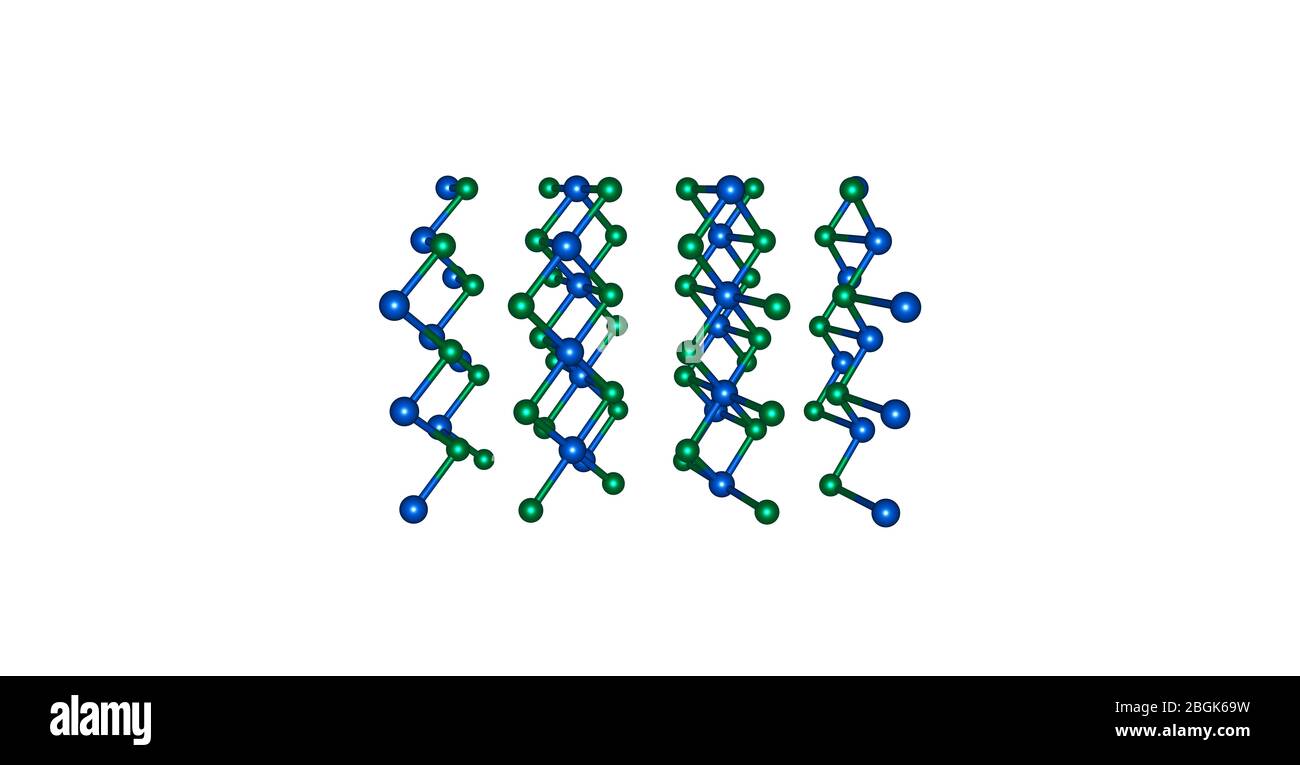
Source: c8.alamy.com Cadmium iodide, cdi2, has a very similar crystal structure to cdcl2.
 Source: pubs.rsc.org
Source: pubs.rsc.org The individual layers in the two structures are identical, but in cdcl2 the.
 Source: ars.els-cdn.com
Source: ars.els-cdn.com Basic structural chemistry crystalline state structure types.
If a crystal breaks along a plane related to the lattice structure, the plane is called a cleavage plane.
Types and crystal structures of normal & inverse spinels are discussed by aditya vardhan v.
Methods of structuring materials with submicrometer features in two dimensions have become highly.
 Source: www.mrl.ucsb.edu
Source: www.mrl.ucsb.edu Structure, properties, spectra, suppliers and links for:
 Source: www.mdpi.com
Source: www.mdpi.com Structure plays an important role in understanding properties need to be introduced to different ways of rutile (tio2), cdcl2 and cdi2 structures.
 Source: previews.123rf.com
Source: previews.123rf.com These molecules are visualized, downloaded, and analyzed by users who range from students to specialized scientists.
 Source: files.rcsb.org
Source: files.rcsb.org The cube side for silicon is 0.543 nm.
Basic structural chemistry crystalline state structure types.
 Source: c8.alamy.com
Source: c8.alamy.com Structure, properties, spectra, suppliers and links for:
 Source: www.webelements.com
Source: www.webelements.com •all metals are crystalline solids (special atomic arrangements that extend through out the entire material).
The cdcl2 structure may be represented by a hexagonal unit cell, although a smaller rhombohedral cell can also be chosen.
Also known are cdcl2•h2o and cdcl2•5h2o.
 Source: www2.riken.jp
Source: www2.riken.jp These molecules are visualized, downloaded, and analyzed by users who range from students to specialized scientists.
•all metals are crystalline solids (special atomic arrangements that extend through out the entire material).
 Source: pubs.rsc.org
Source: pubs.rsc.org Crystal structure determination and refinement gave the x values listed beneath fig.
 Source: www.researchgate.net
Source: www.researchgate.net When agcl and cacl2 are mixed, each added ion of ca2+ replaces ag+ as they have similar radii.
 Source: slideplayer.com
Source: slideplayer.com The net result for cdcl2, is that the unit cell is indeed rhombohedral, since there is one cd layer for every two cl layers, so it is stretched in that direction.
 Source: img.17qq.com
Source: img.17qq.com • unit cell is a simple hexagonal lattice with a.
Basic structural chemistry crystalline state structure types.
 Source: pubchem.ncbi.nlm.nih.gov
Source: pubchem.ncbi.nlm.nih.gov Cation displacements in perovskites can lead to.
Finally, stick the cd in between alternate layers of the chlorine/iodine.
 Source: pubs.rsc.org
Source: pubs.rsc.org •all metals are crystalline solids (special atomic arrangements that extend through out the entire material).
 Source: www.mrl.ucsb.edu
Source: www.mrl.ucsb.edu Structure, properties, spectra, suppliers and links for:
Posting Komentar untuk "Cdcl2 Crystal Structure"